 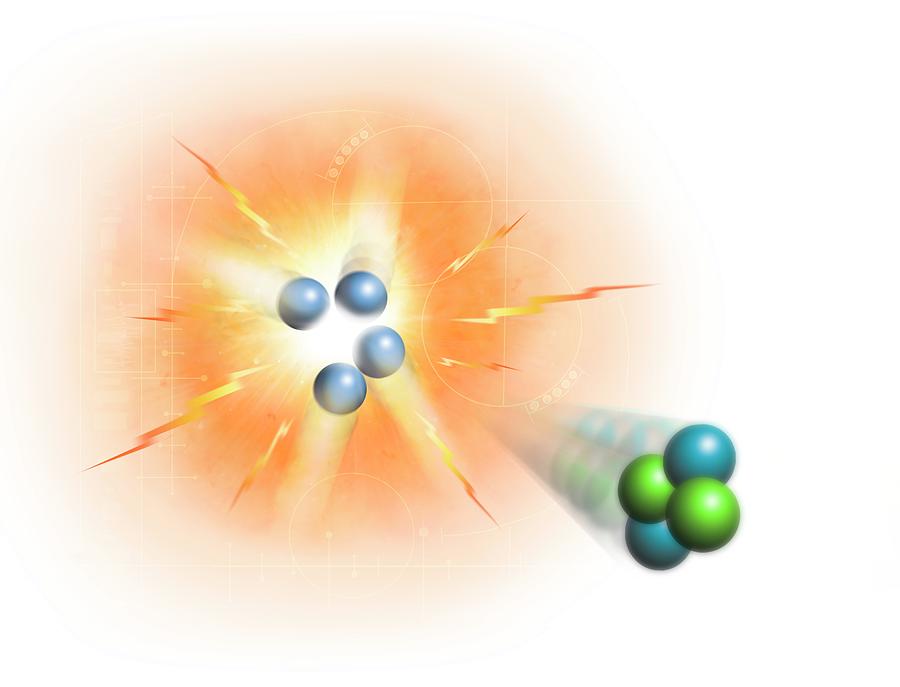
In general, the size of an atom will decrease as you move from left to the right of a certain period. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75. It is colorless, odorless, tasteless, 9 non-toxic, and highly combustible. It is the lightest element and, at standard conditions, is a gas of diatomic molecules with the formula H2. You should know the names and symbols of the more common elements, including those whose symbols are derived from their Latin names. An atom gets larger as the number of electronic shells increase therefore the radius of atoms increases as you go down a certain group in the periodic table of elements. Hydrogen is a chemical element it has symbol H and atomic number 1. The fusion of hydrogen atoms in stars to produce helium produces huge amounts of. The U-236 nucleus then rapidly breaks apart into two smaller nuclei (in this case, Ba-141 and Kr-92) along with several neutrons (usually two or three), and releases a very large amount of energy.Īmong the products of Meitner, Hahn, and Strassman’s fission reaction were barium, krypton, lanthanum, and cerium, all of which have nuclei that are more stable than uranium-235. Make sure you thoroughly understand the following essential ideas: Give a chemical definition of element, and comment on the distinction between the terms atom and element. Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases. Hydrogen atoms all have one electron occupying the space outside of the nucleus. Since atoms are neutral, the number of electrons in an atom is equal to the number of protons. 1: The social security number subatomic-the proton. Nuclear fusion is the process by which two or more atomic nuclei join together to form a single heavier nucleus and large amounts of energy. \): When a slow neutron hits a fissionable U-235 nucleus, it is absorbed and forms an unstable U-236 nucleus. By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties. Lithium atoms have three protons, beryllium atoms have four, and so on. Depiction of the deuterium (D) and tritium (T) fusion reaction, which produces a helium nucleus (or alpha particle) and a high energy neutron.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
